Effects of White-coat Hypertension on Heart Rate Recovery and Blood Pressure Response during Exercise Test
Article information
Abstract
Objectives
White-coat hypertension is defined as high blood pressure (BP) on clinical assessment but normal BP elsewhere or on ambulatory measurement. Autonomic dysfunction may be one of the mechanisms causing white-coat hypertension. Slowed heart rate recovery and excessive BP response during exercise test are associated with autonomic dysfunction. The purpose of this study was to determine the association between white-coat hypertension and abnormal autonomic nervous system response.
Methods
We assessed 295 patients stratified into three groups via 24hr ambulatory BP monitoring, following 2017 ACC/AHA guidelines: normal BP group, white-coat hypertension group, and a hypertension group. We analyzed medical history, blood test, echocardiography, 24hr ambulatory BP monitoring, and exercise test data.
Results
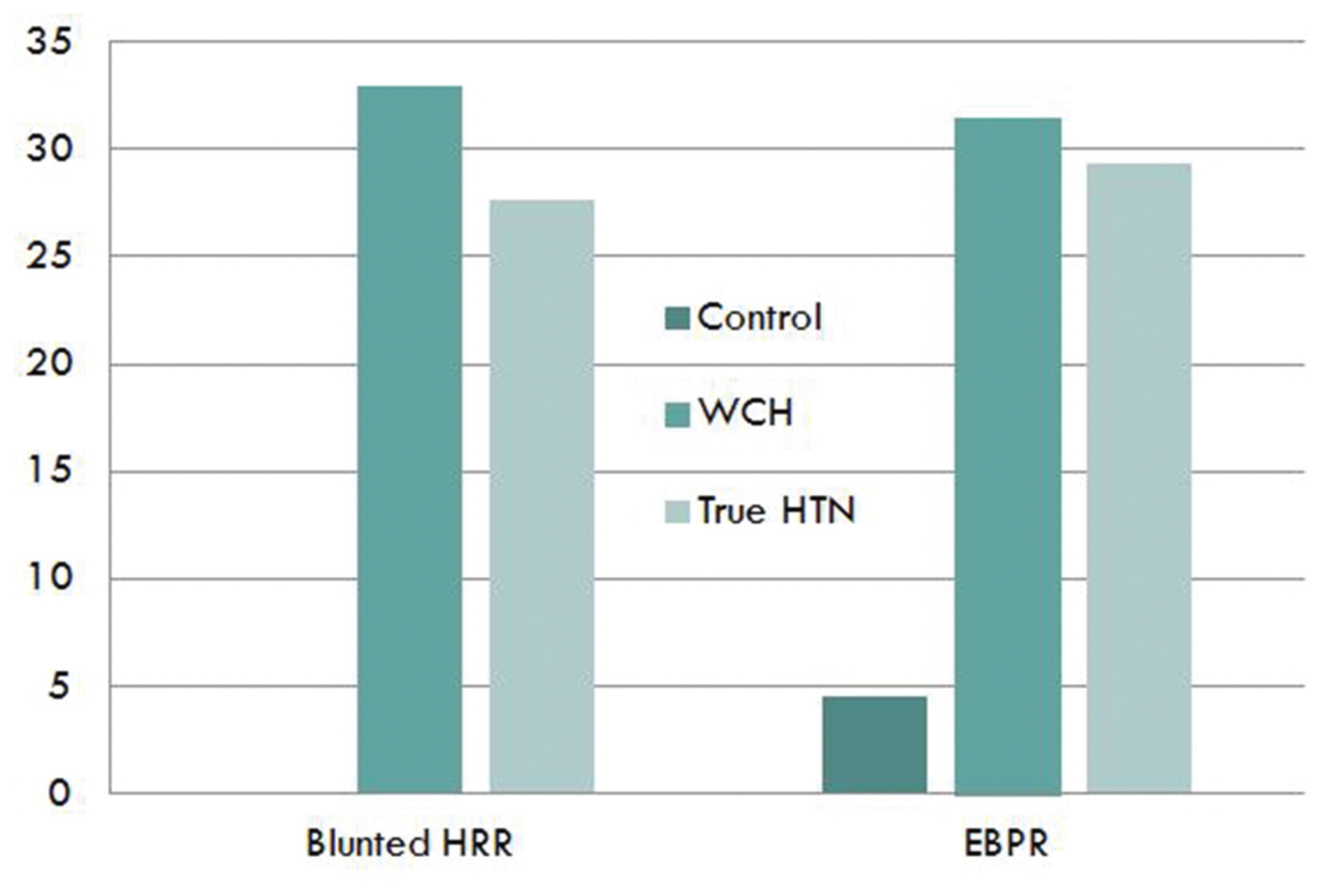
There was no difference in basement characteristics and echocardiography among the groups. Blunted heart rate recovery of each group showed a significant difference. Control group had 0% blunted heart rate recovery, but 33.3% in white coat group and 27.6% in true hypertension group (P < 0.001). Also, in the control group, 4.5% showed excessive BP response, but 31.5% in the white coat hypertension group and 29.3% in the true hypertension group (P < 0.001). Excessive BP response during the exercise test or blunted heart rate recovery, which is an indicator of autonomic nervous system abnormality, was more common in the hypertensive group and white-coat hypertension group than in the normal BP group.
Conclusions
These results confirmed that white-coat hypertension has an autonomic nervous system risk. Therefore, white-coat hypertension can be a future cardiovascular risk factor.
Hypertension is one of the most common diseases that affects about 30% of the population over the age of 30 years in Korea.1 The prevention, diagnosis, and management of hypertension is very important because it is closely related to cerebrovascular disease and cardiovascular disease, which accounts for several deaths in Korean adults.2
Patients with normal blood pressure (BP) during rest may occasionally stop testing their BP level due to excessive elevation in systolic BP (SBP) during exercise testing. One of the underlying mechanisms of the hypertensive response to exercise is the increase in the sympathetic response.3 Increased sympathetic activity is known to be one of the mechanisms underlying the exaggerated BP response to exercise (EBPR).4
EBPR is associated with a higher rate of hypertension than normal, and is associated with increased cardiovascular mortality.5 The risk of hypertension can be confirmed through the exercise test.6 In addition, exercise capacity and heart rate recovery (HRR) were significant predictive factors of the relative risk of death.7 Measurement of HRR is a simple non-invasive procedure analyzing the autonomic nervous dysfunction.8,9 An earlier study showed that a blunted HRR, which is defined as a decrease in heart rate (HR) of less than 12 beats/min from peak exercise to 1 min into recovery, is a powerful predictor of overall mortality.7,10
White-coat hypertension is defined as high BP on clinical assessment but normal BP elsewhere or on ambulatory measurement. In this case, ambulatory BP monitoring and home BP monitoring are recommended.11 Autonomic dysfunction may be one of mechanisms causing white-coat hypertension.12 Its association with HRR and EBPR in white-coat hypertension patients has not been well studied.
We aimed to investigate the association between HRR and EBPR in patients with white-coat hypertension.
MATERIALS AND METHODS
This is a cross-sectional, single center, case control study. We retrospectively reviewed 295 patients who underwent Treadmill test and 24-hour ambulatory BP monitoring (ABPM) between January 2008 and February 2015. Inclusion criteria were: 18–80 years of age, normal renal function, and for women to be on a regular menstrual cycle. Exclusion criteria were: any systemic disease such as significant liver disease, neurologic disorders or malignant disease, or secondary hypertension.
Patients were classified according to the ABPM, following diagnostic criteria suggested by the 2017 ACC/AHA guidelines.13 Hypertension group was meeting one or more of these criteria; a 24-hour mean of 125/75 mmHg or above, daytime (awake) mean of 130/80 mmHg or above or Nighttime (asleep) mean of 110/65 mmHg or above. White coat hypertension is defined as having elevated clinic blood pressure, at or above threshold for hypertension without elevated out-of-office blood pressure, below threshold for hypertension. Patients not included in the two groups were classified as normotensive control. After all, patients were classified as 174 patients with true hypertension, 54 patients with white-coat hypertension, and 67 normotensive controls were included.
Demographic characteristics recorded at the first visit included age, sex, height, weight, current medications, smoking history, and other comorbidities. Blood was drawn for the measurement of total serum cholesterol, high-density lipoprotein (HDL), low-density lipoprotein (LDL) cholesterol, triglycerides, blood glucose, creatinine, uric acid, and high sensitivity C-reactive protein (Hs-CRP). Body mass index (BMI) was calculated as the ratio of weight in kilograms to height in square meters. This study was approved by the Kosin University International Review Board.
- BP measurement and 24-h ambulatory BP monitoring
Office BP measurements were performed twice at 5-min intervals using a mercury sphygmomanometer. Noninvasive 24-h ABPM was performed on each patient’s non-dominant arm using an automatic oscillometric device (TONOPORT V, PAR Medizintechnik, Berlin, Germany) on a normal working day. Patients were asked to refrain from performing fast exercises. All subjects were instructed to rest or sleep between 10:00 PM and 7:00 AM (nighttime) and continue their usual activities between 7:00 AM and 10:00 PM (daytime). The accuracy of the device was checked against the standard auscultatory method of measuring BP to ensure that the difference in BP measurements between methods did not exceed 5 mmHg. The device was set to obtain BP readings at 20-min intervals during the daytime and at 40-min intervals during the nighttime. Only 24-h recordings that included at least 80% successful recordings were accepted as valid. Each ABPM dataset was first automatically scanned to remove artifactual readings according to preselected editing criteria. The following ABPM parameters were evaluated: 24-h mean SBP and diastolic BP (DBP) levels, daytime mean SBP and DBP levels, nighttime mean SBP and DBP levels, and BP variability assessed with standard deviation (SD).
- Treadmill test
All patients underwent symptom-limited exercise stress testing (GE CASE T2100; GE Medical Systems, Milwaukee, WI, USA) according to the protocol by Bruce et al..14 BP was measured using an automated BP monitor (Suntech Tango; Suntech Medical, Morrisville, NC, USA) throughout the treadmill test using the same arm as was used to measure the resting BP. Twelve-lead electrocardiography was monitored continuously and printed at a paper speed of 25 mm/s; measurements of HR and BP were recorded at the end of each 3-min stage at peak exercise and at 1-min and 2-min intervals throughout recovery.
Treadmill test was continued until the participants felt intolerable fatigue or their HR exceeded 95% of estimated maximal HR (220 bpm, age). The total exercise time was also recorded. EBPR was defined as the peak exercise SBP of ≥ 210 mmHg in men and ≥ 190 mmHg in women.15 Functional capacity was estimated in metabolic equivalents (METs) on the basis of speed and grade of the treadmill.16 During the recovery phase, the subjects continued to walk for 60s at a speed of 1.5 mph, and then they sat down for 3min with continued monitoring of BP, HR, and heart rhythm. HRR was defined as peak heart rate minus heart rate after a 1-min recovery; abnormal HRR was defined as ≤ 12 beats/min.17
- Echocardiographic measurement
Standard 2-dimensional echocardiography was performed on all subjects lying in the left lateral decubitus position using a 3.5-MHz transducer (Philips iE33, Philips Medical Systems, Bothell, WA, USA) and the echocardiography examiners were blinded to patient information. Measurements of thickness of the interventricular septum and posterior wall, diameter of the left ventricle (LV) cavity, and the LV mass index (LVMI) were performed according to criteria outlined by the American Society of Echocardiography.18 Pulsed wave Doppler of transmitral LV inflow was performed in the apical four-chamber view with the sample volume placed at the level of the mitral valve tips; doppler variables were analyzed during three consecutive beats. The following measurements of global LV diastolic function were determined: peak early (E) and late (A) diastolic mitral flow velocity, E/A ratio, and early (Ea) diastolic mitral annular velocity.
- Statistical analysis
Statistical analyses were performed using the commercially available computer program SPSS 18.0 for Windows (IBM, Chicago, IL, USA). Data are presented as mean ± standard deviation for continuous variables and percentage (%) if the data are categorical. The Mann–Whitney U test was used for continuous variables and the Chi-square test was used for categorical data. The normality of data was tested using the Kolmogorov–Smirnov test. For all tests, the significance level was set to P < 0.05.
RESULTS
1. Characteristics & Medical history
From January 2008 to February 2015, 295 patients were enrolled in this study. Patients were divided into control, white-coat hypertension, and true hypertension groups.
At baseline, compared to the normotensive control group, the white-coat hypertension group showed higher BMI, SBP, and DBP. BMI was lowest in the control group at 23.2 kg/m2 and highest at 24.9 kg/m2 in the true hypertension group (P = 0.001). SBP/DBP was lowest in the control group and highest in the white-coat group (P < 0.001). There was no difference in age, sex, heart rate, current smoking, diabetes, and dyslipidemia among the groups (Table 1).
There was no statistically significant difference in medical history with regard to aspirin, RAS blocker, calcium channel blocker, diuretics, beta blocker, and statin intake.
2. Laboratory test
In laboratory data, uric acid, eGFR, Hs-CRP, WBC, and Platelet levels were similar between the groups. Mean total cholesterol, LDL cholesterol, HDL cholesterol, and triglycerides were the highest in the true hypertension group, but the difference was not significant.
3. 24-h ambulatory HR & BP monitoring
24-h ambulatory BP monitoring was done. As a result, 24-h heart rate and daytime and night time heart rate were higher in the true hypertensive group than in the control and white hypertensive groups (P = 0.001). Additionally, the 24-hour systolic blood pressure/diastolic blood pressure was 142/91 in the true hypertension group, but 119/75 and 122/75 in the control and white-coat hypertension groups respectively. This represents a significant difference in BP (P < 0.001). Day time SBP, DBP, SBP-SD, DBP-SD, MBP, and MBP-SD showed meaningful differences between control, white-coat hypertension, and true hypertension groups. All results were highest in the true hypertension group, and the same appeared in the night time (P < 0.001) (Table 2).
4. Echocardiography
Echocardiography was performed on all patients and compared according to hypertension type. Ejection fraction (EF), diastolic left ventricular internal dimension (LVIDd), systolic left ventricular internal dimension (LVIDs), left atrium (LA) diameter, LA volume, E velocity, A velocity, and E/Ea shows no significant difference. The white-coat hypertension and true hypertension groups have significantly higher epicardial fat thickness value, and the left ventricle posterior wall diameter on echocardiography was thicker in the true hypertension group than in the control and white hypertensive groups (P < 0.001).
5. Exercise test
Compared to the normotensive group, there is no difference in exercise time, rest heart rate, max heart rate, QT, and QTC. However, HRR was significantly lower in the white-coat hypertension and true hypertension groups when compared to the normotensive group (P < 0.001). HRR between groups showed a significant difference. The control group had 0 blunted HRR, but 33.3% (18 patients) in the white-coat group and 27.6% (48 patients) in the true hypertension group. This shows that the prevalence of blunted HRR was significantly higher in the white-coat hypertension and true hypertension groups than the normotensive group (Table 3).
In the control group, 4.5% (3 patients) showed EBPR, but 31.5% (17 patients) in the white-coat hypertension group and 29.3% (51 patients) in the true hypertension group showed EBPR. The white-coat hypertension group showed the highest percentage of EBPR among all the groups.
In conclusion, the white-coat hypertension group and the true hypertension group showed significantly low HRR and higher prevalence of blunted HRR (Fig. 1).
DISCUSSION
The main finding of our study is that the parameter related with the nervous system responses to exercise such as HRR and EBPR are significantly different in the white-coat hypertension group from the normotensive group. These abnormal responses are similar to that of the true hypertension group. These results could represent another pathomechanism of white-coat hypertension.
The prognostic impact of white-coat hypertension is still a matter of debate and controversy.19 Accumulating evidence focusing on the association of white-coat hypertension with subclinical target organ damage and, more importantly, incident cardiovascular disease suggests that the risk is intermediate between normotension and sustained hypertension. Pierdomenico et al.20 stated that the white-coat hypertension group did not differ significantly from the normotensive group in terms of risk of a cardiovascular event, but because the white-coat hypertension group patients have much greater rate of antihypertensive treatment at follow up than the normotensive group, the results are not conclusive. The risk of cardiovascular event in white-coat hypertension was shown in a study by Verdecchia et al.21 to be dependent on baseline value of BP on ABPM. In this study, data identified that the incidence of cardiovascular events tends to increase consistently above the daytime BP cut-off value of 130/80 mmHg. Similarly, as per the International Database of Home BP in relation to Cardiovascular Outcomes (IDHOCO), among the untreated subjects, cardiovascular risk was significantly higher in the white-coat hypertension group than in the normotensive group.22 Furthermore, a meta-analysis of 14 studies with 29,100 participants performed by Briasoulis et al..23 showed that the incidence of overall cardiovascular events was 6.0% in the white-coat hypertension subjects when compared to 4.0% in the normotensive subjects, thus meaning a 73% increased risk (P < 0.001). The risk for fatal cardiovascular events was increased even more, the incidence being 4.0% and 1.2% in the white-coat hypertension and normotensive groups, respectively.
There are many studies on the identification of the mechanism of white-coat hypertension, but these are not clear. The current study investigates whether individuals with white-coat hypertension have abnormal autonomic-cardiac regulation or impaired vascular function, similar to that observed in sustained or persistent hypertension. Sustained hypertension is associated with sympathetic predominance or diminished parasympathetic nervous system influences on heart rate, or both.3, 24 Such “autonomic dysregulation” may be related to hypertension by increasing cardiac output, vascular resistance, and salt retention. Parasympathetic and sympathetic cardiac nervous system functions can be quantified noninvasively through spectral analysis of variability in heart rate.25 Generally, an increase in HR during exercise occurs as a result of combined sympathetic activation and parasympathetic withdrawal.26 In contrast, parasympathetic reactivation is the principal determinant of decreased HR during early recovery. Given the prognostic significance of diminished parasympathetic tone at rest, post-exercise HRR measurement is a noninvasive procedure that can be used to assess parasympathetic activation.27 HRR is simple to calculate using the data obtained from standard exercise tests; moreover, because it does not require 24-h Holter monitoring or baro-reflex sensitivity testing, HRR may be valuable for assessing the risk in routine clinical practice, including exercise test.
EBPR is also related with the risk of future hypertension and cardiovascular mortality.28 We observed that the white-coat hypertension group had a higher prevalence of EBPR, suggesting that white-coat hypertension might present due to the pathomechanism of impaired vascular response. A recent study by Androulakis el al. showed that the white-coat hypertension group presented with worse endothelial function, more pronounced arterial stiffness, and LVH.29 BP reflects the changes in cardiac output during exercise or recovery that can lead to changes in SBP. Cardiovascular reactivity to both isometric and dynamic exercise has been shown to be one of the most important markers for predicting hypertension. Impaired vascular function, including increased arterial stiffness and abnormal endothelial function, is associated with increased exercise BP response.30 Among the parameters of BP response, exercise BP response is an important marker of cardiovascular risk that is associated with cardiovascular mortality. In particular, EBPR is a significant predictor of cardiovascular events and for new onset of resting hypertension.
This study has several limitations. First, this is a retrospective study. Second, the cross-sectional study design eliminated our ability to determine causal relationships. Third, this study was performed at a single center, and relatively small number of subjects. So it is possible that biases existed with respect to patient referral and population sampling. In order to overcome this limitation, it is thought that additional large randomised control studies will be needed. At last, our patients were treated by various medications that may have had some effect on BP. At base line, the normotensive group patients also were on RAS blockers, beta blockers, or Calcium channel blockers due to cardiovascular disease or renal disease. These medications might have had an impact on the BP variability and HRR.
Our findings suggest that autonomic dysregulation assessed by HRR and vascular dysfunction assessed by EBPR have a proportional relationship with white-coat hypertension like true hypertension. The presence of blunted HRR and EBPR showed significant association in the white-coat hypertension group when compared to the normotension group. This means that autonomic dysregulation and vascular dysfunction could be the pathomechanism of white-coat hypertension, and white-coat hypertension is a risk for a cardiovascular event. Based on these study findings, more attention is to be given to the role of white-coat hypertension in cardiovascular target organ damage, and more study in this area is warranted.