Articles
- Page Path
- HOME > Kosin Med J > Volume 37(2); 2022 > Article
-
Original article
STAT3 inhibition decreases ATP-induced MUC8 gene expression in human airway epithelial cells -
Cheol Hong Kim1
 , Kyoung Seob Song2
, Kyoung Seob Song2
-
Kosin Medical Journal 2022;37(2):134-139.
DOI: https://doi.org/10.7180/kmj.22.102
Published online: June 24, 2022
1Department of Pediatrics, Samsung Changwon Hospital, Sungkyunkwan University School of Medicine, Changwon, Korea
2Department of Medical Science, Kosin University College of Medicine, Busan, Korea
- Corresponding Author: Kyoung Seob Song, PhD Department of Medical Science, Kosin University College of Medicine, 262 Gamcheon-ro, Seo-gu, Busan 49267, Korea Tel: +82-51-990-6655 Fax: +82-51-990-3081 E-mail: kssong@kosin.ac.kr
Copyright © 2022 Kosin University College of Medicine.
This is an open-access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 1,370 Views
- 11 Download
- 2 Crossref
Abstract
-
Background
- Contact between the human pulmonary system and bacteria, viruses, or other pathogens can induce airway diseases. Although pathogen-induced mucus oversecretion and hyperproduction are frequently observed in the human respiratory tract, the molecular mechanisms of pathogen-induced mucus hypersecretion and overproduction remain unclear. The objective of this study was to investigate the physiological signaling mechanism of adenosine triphosphate (ATP)-induced MUC8 gene expression in human airway epithelial cells.
-
Methods
- Real-time reverse transcription polymerase chain reaction, a cytokine array, and a Ca2+ concentration assay were performed to investigate the ATP/P2Y2-induced MUC8 gene expression levels in human airway epithelial cells.
-
Results
- The ATP/P2Y2 complex robustly secreted interleukin (IL)-6 in a time-dependent manner, whereas siRNA-P2Y2 did not. Moreover, ATP/P2Y2 induced MUC8 gene expression. IL-6 secreted by ATP strongly elevated ATP/P2Y2-induced MUC8 gene expression compared to ATP/P2Y2. Interestingly, a specific signal transducer and activator of transcription 3 (STAT3) inhibitor, 5,15-DPP, dramatically inhibited ATP/P2Y2/IL-6-induced STAT3 phosphorylation and resulted in an approximately 5-fold decrease in MUC8 gene expression.
-
Conclusions
- We showed that IL-6-activated STAT6 is essential for ATP/P2Y2-induced MUC8 gene expression as part of inflammatory signaling by cytokines during airway inflammation. Our results provide a new molecular understanding of the signaling mechanism of MUC8 gene expression during airway inflammation.
- Mucus has an essential function in the innate immune system against pathogens such as bacteria, virus, air pollutants, or fungus in the human respiratory track. Mucus hyperproduction and hypersecretion in airway track are frequently observed in a number of respiratory diseases, including asthma, chronic bronchitis, chronic obstructive pulmonary disease, and cystic fibrosis [1]. Various mucins have been named, but their exact physiological functions have yet to be identified. MUC8 has also been known to be expressed in the sinuses of chronic rhinosinusitis patients [2]. The partial cDNA sequence of MUC8 has been identified by Shankar et al. [3]. For this reason, the mechanisms by which MUC8 affect inflammation in the respiratory track are unknown. Recently, we reported that MUC8 acts as anti-inflammatory mucin in reverse using the siRNA-MUC8 system [4]. In this study, we examined the signal mechanism of adenosine triphosphate (ATP)/P2Y2-induced MUC8 gene expression in human airway epithelial cells.
- Purinergic receptors are ubiquitously expressed in many tissues and contribute to innate and adaptive immunity [5,6]. Of purinergic receptors, P2Y2 is expressed in the apical membrane of airway track cells [7] and is an important physiological receptor for airway inflammation [8]. P2Y2 is a Gαq-coupled receptor involved in intracellular signaling by heterotrimeric G proteins. This physiological mechanism has provided information on characterization of the receptor for ATP-induced MUC8 gene expression, its Gαq-protein coupling, and secondary messengers of the downstream process after P2Y2 receptor activation. Thus, understanding of intracellular signaling cascades that trigger mucus overproduction/hypersecretion is required for inflammatory control in an affected microenvironment.
- Recently, a small molecule signal transducer and activator of transcription 3 (STAT3) inhibitor, C188-9, prevented house dust mite-induced airway remodeling, airway inflammation, and T helper type 2 and 17 (Th2/Th17) cells accumulation [9]. This was not surprising because local blockade of interleukin (IL)-6 decreased STAT3 activation, increased Th17 inflammation, and increased Th2 responses in mice [10,11]. However, the biochemical and physiological mechanisms by which ATP/P2Y2-induced MUC8 gene expression is affected by IL-6 in respiratory diseases remain unclear.
Introduction
- 1. Materials and cell culture
- ATP and 5,15-DPP were purchased from Merck (Darmstadt, Germany). The IL-6 ELISA kit was obtained from R&D Systems (Minneapolis, MN, USA). siRNA specifically targeting P2Y2 was synthesized by Bioneer (Daejeon, Korea). The siRNA sequences of P2Y2 were GAGGAAGGUGGCUUACCAA (dTdT). The human lung mucoepidermoid carcinoma cell line (NCI-H292) was obtained from the American Type Culture Collection (CRL-1848; Manassas, VA, USA). Cells were incubated in RPMI 1640 (Invitrogen, Carlsbad, CA, USA) added with 10% fetal bovine serum in the presence of penicillin/streptomycin at 37°C in a humidified chamber with 5% CO2.
- 2. Real-time RT-PCR
- Real-time reverse transcription polymerase chain reaction (RT-PCR) was performed using a Bio-Rad iQ iCycler Detection System (Bio-Rad Laboratories, Hercules, CA, USA) with iQ SYBR Green SuperMix. The following primers were used: MUC8, forward (5’-GACCTGCCCCCATGGAC-3’) and reverse (5’-CAGGAGTTCGAGACCAGCCT-3’). β2M, forward (5’-CGCTCCGTGGCCTTAGC-3’) and reverse (5’-GAGTACGCTGGATAGCCTCCA-3’). Reactions were performed in a total volume of 20 µL, which included 10 µL of 2× SYBR Green PCR Master Mix (Thermo Fisher, Waltham, MA, USA), 300 nM of each primer, and 1 µL of previously reverse-transcribed cDNA template. Real-time RT-PCR was performed on a MiniOption Real-time RT-PCR Detection System (Bio-Rad Laboratories). The parameters were 95°C for 10 minutes, followed by 40 cycles of 95°C for 15 seconds, 60°C for 30 seconds, and 72°C for 30 seconds. All reactions were performed in triplicate. The relative quantity of mRNA was obtained using the comparative cycle threshold method and was normalized using β2-microglobulin as an endogenous control [12].
- 3. Cytokine assay
- Cytokine levels were quantified using a Human Cytokine Array Panel A kit (R&D Systems, ARY005B) according to the manufacturer’s instructions. Briefly, cells were plated in six-well plates 1 day before transfection with either a construct driving the expression of P2Y2 or P2Y2-specific siRNA using FuGENE 6 (Roche, Indianapolis, IN, USA). Twenty-four hours after transfection, serum-starved cells were treated with ATP for 4 hours. After treatment, supernatants were assayed for cytokine production according to the kit’s instructions [13].
- 4. Calcium colorimetric assay
- Ca2+ concentration level was quantified using a Calcium Colorimetric assay kit (ab102505; Abcam, Cambridge, UK) according to the manufacturer’s instructions. Briefly, cells were plated in six-well plates 1 day before transfection with a construct driving the expression of P2Y2 using FuGENE 6 (Promega, Madison, WI, USA). Twenty-four hours after transfection, serum-starved cells were treated with ATP for 1 hour. The cells were assayed for Ca2+ concentration according to the kit’s instructions.
- 5. Statistical analysis
- The data are presented as the mean±standard deviation of more than three independent experiments. When appropriate, statistical differences were measured using Wilcoxon Mann-Whitney tests. A p-value less than 0.05 was considered statistically significant.
Methods
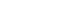
- 1. ATP activates P2Y2 purinergic receptor to induce MUC8 gene expression in NCI-H292 cells
- We performed real-time RT-PCR to determine whether P2Y2 receptor is essential for ATP-induced MUC8 gene expression in human airway epithelial (NCI-H292) cells. After transfection with either wild-type P2Y2 or siRNA-P2Y2 construct, ATP was applied for 24 hours (Fig. 1A). The ATP/P2Y2 complex dramatically induced MUC8 gene expression compared to ATP but not to siRNA-P2Y2. This result suggests that P2Y2 is critical for ATP-induced physiological functioning. ATP is an inducer of inflammation in many tissues in humans. We posit that ATP signaling can breakdown the secretagogues in cells to augment the inflammatory signal by secreting several cytokines/chemokines from the cells. To test that, cytokine array was performed with cell medium (Fig. 1B). Interestingly, IL-6 was secreted out of the cells by the ATP/P2Y2 complex but not by siRNA-P2Y2, suggesting that P2Y2 mediates ATP-induced IL-6 secretion. In addition, IL-6 secretion peaked at 6 hours after ATP treatment and then decreased. These results suggest that ATP/P2Y2 could result in extracellular secretion of IL-6 to induce an inflammatory condition in airway epithelial cells.
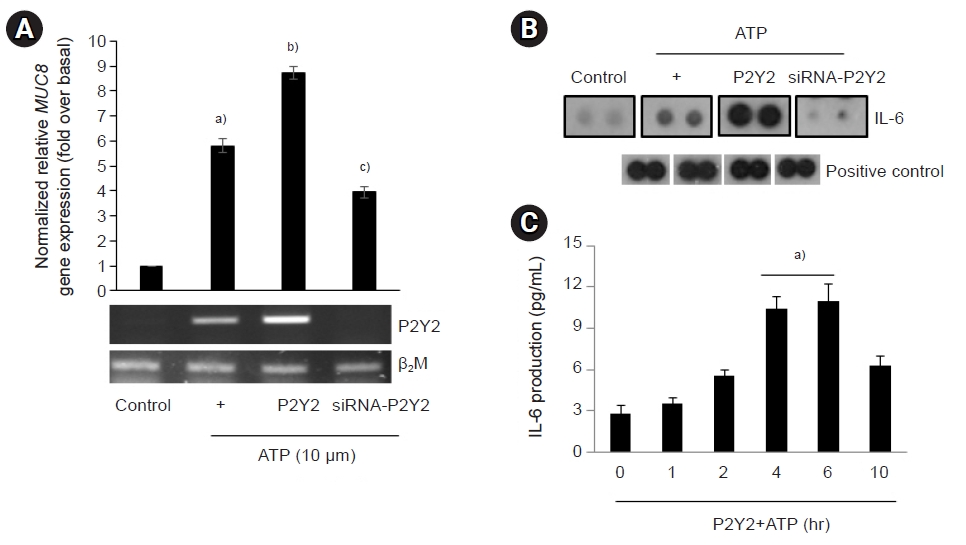
- 2. Secreted IL-6 augments ATP/P2Y2-activated [Ca2+] to increase MUC8 gene expression
- To examine whether secreted IL-6 could affect ATP as an essential factor in P2Y2-induced MUC8 gene expression, extracellular ATP and purified IL-6 were applied to cells transfected with P2Y2 (Fig. 2A). Extracellular IL-6 treatment could strongly increase ATP/P2Y2-treated MUC8 gene expression compared to ATP/P2Y2 only. Because P2Y2 is a Gαq-coupled receptor, intracellular calcium was measured after treatment with IL-6. As expected, intracellular calcium concentration was increased by extracellular IL-6 treatment (Fig. 2B). These results suggest that either/both autocrine or/and paracrine IL-6 can activate intracellular calcium concentration to increase ATP/P2Y2-induced MUC8 gene expression in NCI-H292 cells.
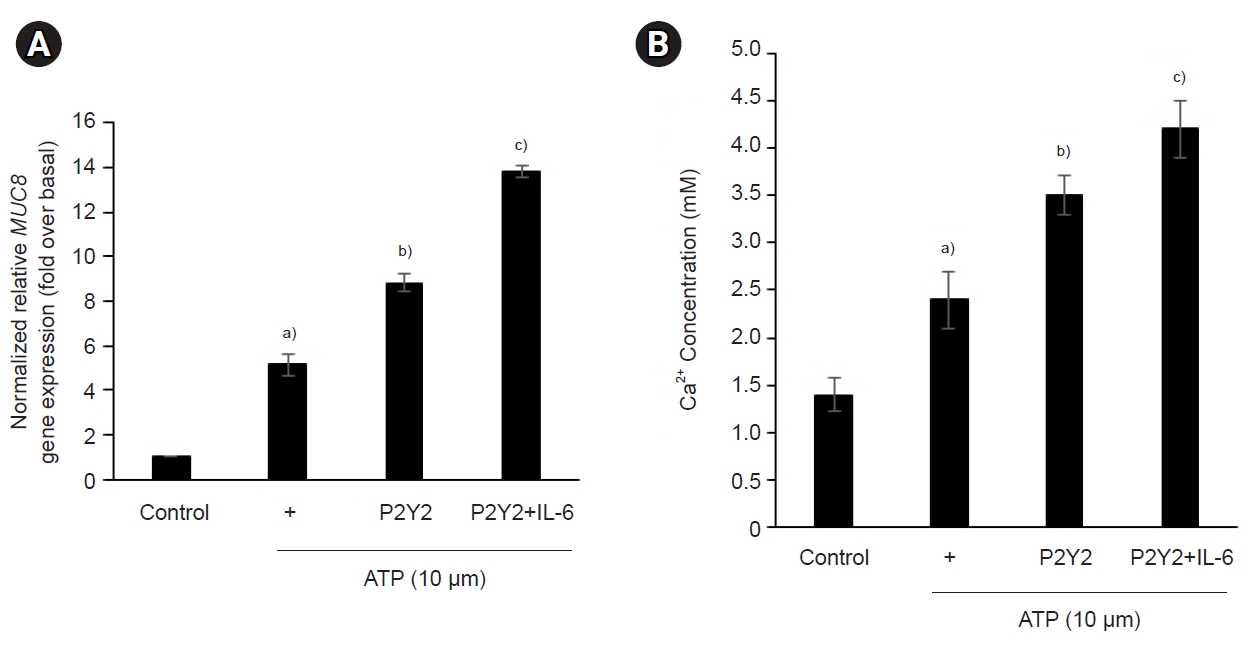
- 3. STAT3 is essential for ATP/P2Y2-induced MUC8 gene expression after cotreatment with IL-6
- To investigate the signaling mechanism of ATP/P2Y2-induced MUC8 overproduction after cotreatment with IL-6, the specific STAT3 inhibitor 5,15-DPP was utilized (Fig. 3). Because STAT3 can be frequently activated by IL-6, it is a pivotal transcription factor for cell proliferation. Interestingly, 5,15-DPP significantly decreased ATP/P2Y2-induced MUC8 in a dose-dependent manner after cotreatment with IL-6. This is why IL-6 secretion is critical for airway inflammation in NCI-H292 cells. Although ATP activated Gαq-coupled receptor and secondary messengers like intracellular calcium, STAT3 might play a critical role in this signal complex.
Results
- We identified how ATP signaling could induce MUC8 gene expression via the P2Y2 G-protein coupled receptor receptor in human airway epithelial cells. Though ATP is an inflammation inducer, the precise signaling mechanism of ATP/P2Y2-induced MUC8 gene expression remains unclear. Interestingly, IL-6 could be secreted extracellularly by the ATP/P2Y2 complex in a time-dependent manner (Fig. 1C) but not by siRNA-P2Y2. IL-6 is a pro-inflammatory cytokine that induces a number of physiological functions. The IL-6 secreted by the ATP complex was uptaken into nearby cells to transfer inflammatory signals. Surprisingly, IL-6 robustly induced ATP/P2Y2-induced MUC8 gene expression compared to ATP/P2Y2 treatment (Fig. 2A). Thus, we posited that ATP could transmit inflammatory signals to boost or magnify inflammatory signaling to nearby cells. More interestingly, siRNA-P2Y2 did not induce this phenomenon, because P2Y2 is an essential ATP purinergic receptor in airway epithelial cells. In fact, many cytokines/chemokines were secreted in an ATP-time/dose-dependent manner, including regulated upon activation, normal T cell expressed, and secreted, IL-1α, and IL-1β; however, only IL-6 was affected by siRNA. Thus, we posit that IL-6 plays a critical role(s) in ATP signaling transduction in the airway.
- Our previous study tested MUC8 as an anti-inflammatory mucin using siRNA study [4]. After 24 hours of ATP treatment, increased MUC8 significantly inhibited ATP/P2Y2-dependent inflammatory cytokine production for more than 72 hours. Additional information for full-length cDNA and promoter sequences of MUC8 is required to support the assumption that MUC8 is an anti-inflammatory mucin.
- IL-6 is a small glycoprotein (21 KDa) produced by cells in the innate immune system [14]. Increased level of IL-6 has been frequently observed in asthmatic patients [15]. More importantly, a study examining IL-6 in bronchoalveolar lavage fluid has shown increased level of IL-6 in active asthmatic patients compared with the levels in healthy nonsmoker, stable asthmatic, and non-asthmatic patients [16]. Thus, recently, scientists reported that inhibition of STAT3, a major downstream signal of IL-6, significantly decreased lung inflammation, airway remodeling, and inflammatory cell infiltration in asthma [9,17-19]. However, there is little information on the role of STAT3 in mucus hypersecretion and overproduction during airway inflammation.
- In summary, our results showed that IL-6 is essential for ATP/P2Y2-induced MUC8 gene expression in human airway epithelial cells. In addition, activation of STAT3 is mainly related to MUC8 gene expression through induction of ATP/P2Y2. We suggest that the mucus hypersecretion/overproduction observed during airway inflammation is a consequence of ATP/P2Y2-induced increase in MUC8 gene expression within the inflamed microenvironment. Further analysis of the mucus hypersecretion/overproduction induced by various cytokines, bacterial products, or viruses might increase understanding of the pathogenesis of respiratory diseases.
Discussion
-
Conflicts of interest
Kyoung Seob Song is an editorial board member of the journal but was not involved in the peer reviewer selection, evaluation, or decision process of this article. No other potential conflicts of interest relevant to this article were reported.
-
Funding
This study was supported by a grant from the National Research Foundation of Korea (NRF), funded by the Korean government (NRF-2020R1I1A2075001 and NRF-2021R1A4A1031380 to KSS).
-
Author contributions
Conceptualization: CHK, KSS. Methodology: CHK, KSS. Investigation: CHK, KSS. Funding acquisition: KSS. Writing-original draft: CHK, KSS. Writing - review & editing: CHK, KSS. Approval of final manuscript: all authors.
Article information
- 1. Kim YO, Jung MJ, Choi JK, Ahn do W, Song KS. Peptidoglycan from Staphylococcus aureus increases MUC5AC gene expression via RSK1-CREB pathway in human airway epithelial cells. Mol Cells 2011;32:359–65.ArticlePubMedPMCPDF
- 2. Lee HM, Kim DH, Kim JM, Lee SH, Hwang SJ. MUC8 mucin gene up-regulation in chronic rhinosinusitis. Ann Otol Rhinol Laryngol 2004;113:662–6.ArticlePubMedPDF
- 3. Shankar V, Pichan P, Eddy RL Jr, Tonk V, Nowak N, Sait SN, et al. Chromosomal localization of a human mucin gene (MUC8) and cloning of the cDNA corresponding to the carboxy terminus. Am J Respir Cell Mol Biol 1997;16:232–41.ArticlePubMed
- 4. Cha HJ, Jung MS, Ahn do W, Choi JK, Ock MS, Kim KS, et al. Silencing of MUC8 by siRNA increases P2Y₂-induced airway inflammation. Am J Physiol Lung Cell Mol Physiol 2015;308:L495–502.ArticlePubMed
- 5. Eltzschig HK, Sitkovsky MV, Robson SC. Purinergic signaling during inflammation. N Engl J Med 2012;367:2322–33.ArticlePubMedPMC
- 6. Schenk U, Westendorf AM, Radaelli E, Casati A, Ferro M, Fumagalli M, et al. Purinergic control of T cell activation by ATP released through pannexin-1 hemichannels. Sci Signal 2008;1:ra6.ArticlePubMed
- 7. Jeong JY, Kim J, Kim B, Kim J, Shin Y, Kim J, et al. IL-1ra secreted by ATP-induced P2Y2 negatively regulates MUC5AC overproduction via PLCβ3 during airway inflammation. Mediators Inflamm 2016;2016:7984853.ArticlePubMedPMCPDF
- 8. Vanderstocken G, Van de Paar E, Robaye B, di Pietrantonio L, Bondue B, Boeynaems JM, et al. Protective role of P2Y2 receptor against lung infection induced by pneumonia virus of mice. PLoS One 2012;7:e50385.ArticlePubMedPMC
- 9. Gavino AC, Nahmod K, Bharadwaj U, Makedonas G, Tweardy DJ. STAT3 inhibition prevents lung inflammation, remodeling, and accumulation of Th2 and Th17 cells in a murine asthma model. Allergy 2016;71:1684–92.ArticlePubMedPDF
- 10. Doganci A, Eigenbrod T, Krug N, De Sanctis GT, Hausding M, Erpenbeck VJ, et al. The IL-6R alpha chain controls lung CD4+CD25+ Treg development and function during allergic airway inflammation in vivo. J Clin Invest 2005;115:313–25.ArticlePubMedPMC
- 11. Finotto S, Eigenbrod T, Karwot R, Boross I, Doganci A, Ito H, et al. Local blockade of IL-6R signaling induces lung CD4+ T cell apoptosis in a murine model of asthma via regulatory T cells. Int Immunol 2007;19:685–93.ArticlePubMed
- 12. Kim JI. Analysis of an EGFR mutation by PNA clamping method in lung carcinoid tumors. Kosin Med J 2015;30:141–7.ArticlePDF
- 13. Kong EH, Ma SY, Jeong JY, Kim KH. Effects of L-ascorbic acid on the production of pro-inflammatory and anti-inflammatory cytokines in C57BL/6 mouse splenocytes. Kosin Med J 2015;30:41–9.ArticlePDF
- 14. Rincon M, Irvin CG. Role of IL-6 in asthma and other inflammatory pulmonary diseases. Int J Biol Sci 2012;8:1281–90.ArticlePubMedPMC
- 15. Yokoyama A, Kohno N, Fujino S, Hamada H, Inoue Y, Fujioka S, et al. Circulating interleukin-6 levels in patients with bronchial asthma. Am J Respir Crit Care Med 1995;151:1354–8.ArticlePubMed
- 16. Tillie-Leblond I, Pugin J, Marquette CH, Lamblin C, Saulnier F, Brichet A, et al. Balance between proinflammatory cytokines and their inhibitors in bronchial lavage from patients with status asthmaticus. Am J Respir Crit Care Med 1999;159:487–94.ArticlePubMed
- 17. Simeone-Penney MC, Severgnini M, Tu P, Homer RJ, Mariani TJ, Cohn L, et al. Airway epithelial STAT3 is required for allergic inflammation in a murine model of asthma. J Immunol 2007;178:6191–9.ArticlePubMed
- 18. Kasembeli MM, Bharadwaj U, Robinson P, Tweardy DJ. Contribution of STAT3 to inflammatory and fibrotic diseases and prospects for its targeting for treatment. Int J Mol Sci 2018;19:2299.ArticlePubMedPMC
- 19. Schaunaman N, Dimasuay KG, Kraft M, Chu HW. Tollip interaction with STAT3: a novel mechanism to regulate human airway epithelial responses to type 2 cytokines. Respir Res 2022;23:31.ArticlePubMedPMCPDF
References
Figure & Data
References
Citations

- Inhibition of Urban Particulate Matter-Induced Airway Inflammation by RIPK3 through the Regulation of Tight Junction Protein Production
Sun-Hee Park, Hyun-Chae Lee, Hye Min Jeong, Jeong-Sang Lee, Hee-Jae Cha, Cheol Hong Kim, Jeongtae Kim, Kyoung Seob Song
International Journal of Molecular Sciences.2023; 24(17): 13320. CrossRef - PDZ Peptide of the ZO-1 Protein Significantly Increases UTP-Induced MUC8 Anti-Inflammatory Mucin Overproduction in Human Airway Epithelial Cells
Han Seo, Hyun-Chae Lee, Ki Chul Lee, Doosik Kim, Jiwook Kim, Donghee Kang, Hyung-Joo Chung, Hee-Jae Cha, Jeongtae Kim, Kyoung Seob Song
Molecules and Cells.2023; 46(11): 700. CrossRef

 KOSIN UNIVERSITY COLLEGE OF MEDICINE
KOSIN UNIVERSITY COLLEGE OF MEDICINE
 PubReader
PubReader ePub Link
ePub Link Cite
Cite